Aggregation Effects on Pigment Coatings: Pigment Red 179 as a Case Study
DOI: 10.1021/acsomega.9b02819
American Chemical Society
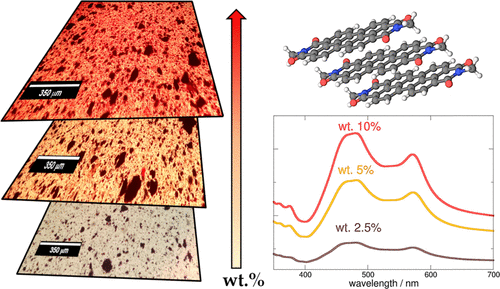
Here, we have studied, with a combined experimental and computational approach, the effect of the crystal environment and aggregation on the electronic properties of Pigment Red 179, which affect both its color and optical energy gap. Spectra acquired in the near-infrared and visible range of energies suggest that this molecule is indeed a “cool” dye, which can be employed as a red pigment that provides effective color coverage to different substrates without contributing to their heating during light irradiation. Spectra acquired on different polymer mixtures at different pigment concentrations (i.e., 2.5–10 wt %) suggest that absorption features depend on chromophoric arrangements promoted by the strong intermolecular π–π interactions. Calculations, performed at the time-dependent density functional theory level, allowed to both attribute the nature of the electronic transitions causing the observed spectra involved and understand the effect of the environment. Indeed, the visible spectra of the pigment is dominated by two localized transitions, with negligible charge transfer for both a dye monomer and dimer either in vacuum or acetonitrile solution. Instead, models including the crystal environment of the pigment show the presence of a high-wavelength S1 ← S0 charge transfer transition between two adjacent molecules, in quantitative agreement with the experimental absorption energy of the crystal pigment.