Conduction Mechanisms in Oxide-Carbonate Electrolytes for SOFC: Highlighting the Role of the Interfa
The Journal of Physical Chemistry C 2018
DOI: 10.1021/acs.jpcc.8b02174
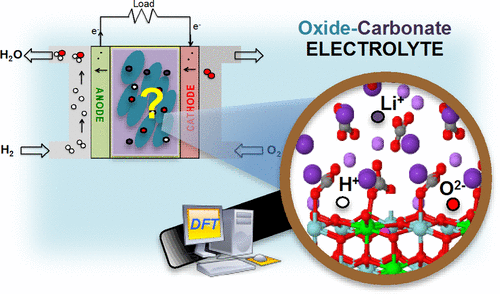
A comprehensive density functional theory investigation of conduction mechanisms of both intrinsic and extrinsic species found in oxide–carbonate composite electrolyte materials used in solid oxide fuel cell is presented. An interface model of yttria-stabilized zirconia–LiKCO3 is used as a case study to investigate transport properties, considering different mechanistic assumptions suggested in the literature over the years. Results clearly indicate that interfaces play an extremely important role in influencing the ionic conductivity performances of these electrolytes. In particular, redistribution of ions of both phases can lead to the formation of the so-called space-charge layer at the interface, which then favorably influences the transport of intrinsic cations or extrinsic protons in particular. These results constitute an important first step toward the understanding of the electrochemical processes and transport mechanisms in these electrolytes, to elucidate the origin of their enhanced conductivity at low temperature when compared to that of their parent components.